Categories
Addiction & DetoxApollo 11 Lunar Mission
Bacteria
Chakra Balance
Chronic Fatigue
Common Cold
Dental Health
Electromagnetic Energy
Epstein-Barr Virus
Fungi
Galen Hieronymus
Hepatitis Virus
Herpes Virus
HIV Virus
Hormonal Imbalance
Influenza Virus
Instructional Videos
Introductory Articles
Lyme Disease
Malaria
Media
Minerals
Miracles
Natural Phenomena
Neurological Disorders
Neurotransmitters
Nutrition
OM Prakash Network HIV Testimonials
Pathogen Cleanse
PCR Test Results
Pet Health
Photographic Force Field
Programs
Quantum Diseases
Scalar Light Scientists
Shared
Shingles Virus
Short Explainer Videos
Superbugs
Testimonials and Case Studies
Uncategorized
Video Testimonials
Viruses
Weight Loss
PCR Testing for Pathogens Relevant to the Scalar Light Pathogenic Cleanse

The scalar light pathogenic cleanse disassembles pathogens while leaving the antibodies untouched and capable of providing defense against pathogenic infection. An antibody, also known as an immunoglobulin, is a protein that is a natural defense mechanism that identifies and neutralizes a pathogen. Antibodies are not treated by way of scalar light and hence will remain inside the body after the pathogen has been disassembled.
I highly recommend a polymerase chain reaction (PCR) test as opposed to an antibody test in order to ascertain the efficacy of a scalar light pathogenic cleanse. A PCR test will diagnose the presence of the genetic material of a pathogen, whereas, an antibody test will diagnose the presence of an antibody that is associated with a particular pathogenic infection. The scalar light pathogenic cleanse disassembles pathogens, while leaving antibodies untouched in order to continue to protect the body against infection. Hence, a PCR test is the most relevant diagnostic test to ascertain whether a pathogen has been disassembled and eliminated by way of a scalar light pathogenic cleanse.
A polymerase chain reaction (PCR) test is an amplification method whereby a tiny amount of DNA that may be present in blood, hair or tissues, can be rapidly copied so that there are ultimately millions of copies of a specific piece of DNA produced for diagnosis. Hence, PCR testing facilitates the detection of DNA of various pathogenic organisms, such as, viruses, bacteria, protozoan and fungi, present inside the human body.
In explanation, a PCR test is a diagnostic method in which a strand of DNA obtained from the blood or another type of biological sample is amplified and replicated through a series of temperature cycles. In order to amplify a segment of DNA using PCR, the sample is first heated so that the DNA denatures, or separates into two pieces of single-stranded DNA. Then, the enzyme Taq polymerase synthesizes two (2) new strands of DNA, using the original strands as templates. Subsequently, this process results in the duplication of the original DNA, with each of the new molecules containing one old and one new strand of DNA whereby these strands can be used subsequently to create two (2) new copies. The cycle of denaturing and synthesizing new DNA is repeated as many as 30 - 40 times thereby producing more than 1 billion exact copies of the original DNA sequence.
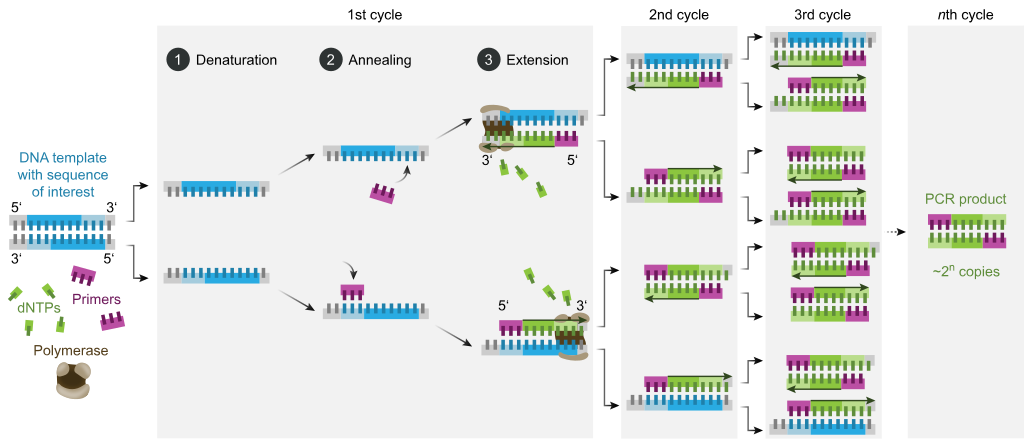
Image from Wikipedia
PCR diagnostic tests are readily available throughout the medical community and should be utilized as an important diagnostic tool in the identification of pathogens. PCR tests are superior to antibody diagnostic tests in that the presence of pathogens can be immediately identified by PCR analysis. Whereas, serologically-based diagnostic tests rely upon the presence of antibodies in response to a pathogenic infection. On many occasions, there is a span of a few weeks between the onset of a pathogenic infection and the eventual antibody response.
Furthermore, a PCR-based test is so exact that it is possible to quantify the amount of a virus in a person's blood, known as the "viral load," thereby providing insight as to the progression of a pathogenic infection as well as the response to therapy. Viral load assessment before, during and after therapy has tremendous potential for improving the clinical management of pathogenic diseases caused by viral infection, including AIDS, herpes, influenza, cytomegalovirus, hepatitis, human metapneumovirus, rhinovirus, respiratory syncytial virus, etc.
The scalar light pathogenic cleanse disassembles pathogens while leaving the beneficial antibodies untouched and capable of providing defense against present and subsequent infection. With this thought in mind, an antibody test after a scalar light pathogenic cleanse may prove to be inconclusive as the infecting pathogen may have been disassembled already by way of scalar light.
With this thought in mind, I recommend a PCR test in order to test exclusively for the presence of a pathogen and not the presence of an antibody or immunoglobulin. With the advent of scalar light treatments it will become necessary to rely upon PCR testing in order to ascertain whether or not the infectious pathogen has been disassembled.
Last, the medical community should always be respected as any accepted diagnostic test has merit and serves a purpose in the treatment of pathogenic disease. My point is to raise awareness as to the accuracy of a PCR test in ascertaining either the presence or absence of a pathogen subsequent to a successful scalar light pathogenic cleanse. May God grant you excellent health. Thank you.